 
Nickel-cadmium batteries are commonly used as rechargeable batteries.However, they have a limited life, are heavy to ship, and contain toxic materials. Lead-acid batteries are reliable sources which provide high currents.The Nernst equation can be used to determine the total voltage, or electromotive force, for a full electrochemical cell (a battery).When a battery is supplying electric power, its positive terminal is the cathode and its negative terminal is the anode. 
Physics Question Pack Passage 9 Question 55 During charging, the anode material is oxidized and produces while the cathode is reduced and consumes electrons. As with all batteries, rechargeable batteries consist of an anode, a cathode, and an electrolyte. Nickel-cadmium (NiCd) batteries, nickel-metal hydride (NiMH) batteries, lithium-ion (Li-ion) batteries, and lithium-ion polymer (LiPo) batteries are a few commonly used rechargeable batteries. 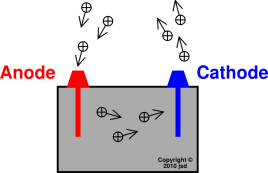
Lead-acid batteries are affected by temperature and must be maintained in order to achieve maximum life expectancy. Depending on the power conversion technology incorporated, batteries can go from accepting energy to supplying energy instantaneously. Lead-acid batteries have moderate power density and good response time. A lead storage battery is the oldest type of rechargeable battery and one of the most common energy storage devices.Īlthough the batteries are reliable, they have a limited life, are heavy to ship, and contain toxic materials that require specific removal methods at the end of their useful life. Terminal voltage is the voltage output of a device measured across its terminals.Ī lead storage battery, also known as a lead-acid provides high currents and stores charge for long periods of time, making them essential for vehicles. Where the uppercase letters are concentrations, and the lowercase letters are stoichiometric coefficients for the overall redox reaction: aA + bB → cC + dDĮlectromotive force (emf) is the potential difference of a source, or a battery, when no current is flowing. – n is the number of moles of electrons transferred in the reaction – Eo is the electromotive force of the standard cell – E is the electromotive force of the non-standard cell The equation relates the electromotive force (emf) of a nonstandard cell to the standard electrode potential, temperature, and activities (often approximated by concentrations) of the chemical species undergoing reduction and oxidation. The Nernst Equation enables the determination of cell potential under non-standard conditions. The Nernst equation can be used to determine the total voltage, or electromotive force, for a full electrochemical cell. All rights reserved.A battery is a device consisting of one or more electrochemical cells with external connections for powering electrical devices. /battery-56a128375f9b58b7d0bc8c92.gif)
This process does not release air/soil pollutants which are usually associated with high temperature pyrotechnic process.Ĭopyright © 2011 Elsevier B.V. Also thermal investigation reveals Pb deposited at Ti-cathode is superior to that from TSIA cathode. The insoluble lead oxides accumulated at the central compartment in the ratio 1:3, respectively for the high oxygen over-voltage Ti-anode (Ti-EK cell) and low oxygen over-voltage TSIA-anode (TSIA-EK cell) shows the superiority of Ti anode over TSIA anode. XRD reveals that the sludge is a mixture of (PbO)(4), Pb(2)O(3), PbSO(4), Pb(S(2)O(3)) and Pb(2)(SO(4)) which upon DC voltage application in a EK cell employing either titanium electrodes or titanium substrate insoluble anode as electrodes caused migration of sulphates and lead ions respectively into anode and cathode compartments, and accumulation of insoluble lead oxides at the central compartment. A novel electrokinetic (EK) technique is applied to separate lead and sulphate from the sludge of used/spent lead acid battery. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
